|
An interactive periodic table may be found here. \): The modern version of the periodic table. 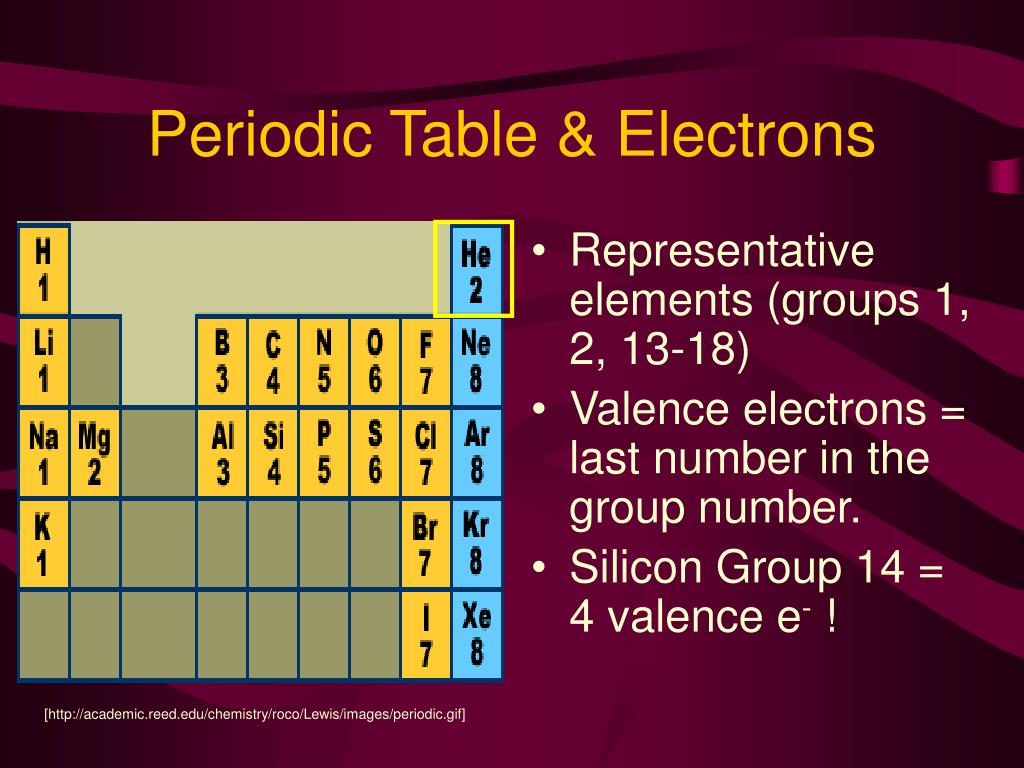
(2012, December 18) Valence Electrons and the Periodic Table. In general, the number of valence electrons is the same within a column and increases from left to right within a row. If the valence shell of an element is full, such as with a noble gas, then the element does not want to gain or lose an electron.įor example, alkali metals, which all have a valency of 1, want to lose that one electron and are likely to form ionic bonds (such as in the case of NaCl, or table salt) with a Group 17 element, which has a valency of 7 and wants to gain that one electron from the alkali metal (Group 1 element) to form a stable valency of 8.įor more on valence electrons and how they're related to the periodic table, I strongly recommend this video:Ĭitations: Tyler Dewitt. For facts, physical properties, chemical properties, structure and atomic properties of the specific element, click on the element symbol in the below periodic table. Transcribed image text: Give the chemical symbol of an element in the third period (row) of the periodic table with three valence electrons. Periodic Table of Elements with Valence Electrons Trends In the below periodic table you can see the trend of Valence Electrons. 
They determine how "willing" the elements are to bond with each other to form new compounds. 98 (57 ratings) Part A: The chemical symbol of an element in the third period of the periodic table with three valence electrons is: Al Explanation: The eleme. Valence electrons are responsible for the reactivity of an element. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table.įor example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively.Ītoms in Groups 13 and 18 have 3 and 8 valence electrons, respectively. Valence electrons are the electrons present in the outermost shell of an atom. By using the electron configuration (condensed notation) of an element, all the electrons outside of the nobel gas core represent the valence electrons. To form a covalent bond, one electron from the halogen and one electron from another atom form a shared pair.įor example, in #"H–F"#, the dash represents a shared pair of valence electrons, one from #"H"# and one from #"F"#. To form an ionic bond, a halogen atom can remove an electron from another atom in order to form an anion (e.g., #"F"^"-", "Cl"^"-"#, etc.). So how does the periodic table help me figure out how. In their chemical reactions halogen atoms achieve a valence shell octet by capturing or borrowing the eighth electron from another atom or molecule. For this reason, the valence electrons are the electrons that are involved in chemical bonding. They have one less electron configuration than a noble gas, so they require only one additional valence electron gain an octet. The halogens (F, Cl, Br etc.) are one electron short of a valence shell octet, and are among the most reactive of the elements (they are colored red in this periodic table). The most reactive nonmetals are the halogens, e.g., #"F"# and #"Cl"#. One simple piece of evidence for this is the Noble Gases which form the last column on the right of the periodic table. Valence Electrons Of Lanthanides And Actinides (Transition And Inner Transition Elements) Consider Scandium (Sc) with its atomic number of 21. Nonmetals tend to attract additional valence electrons to form either ionic or covalent bonds. Chemical elements in the same group of the Periodic Table possess similar valence states and other physico-chemical properties. They need to lose only one or two valence electrons to form positive ions with a noble gas configuration. The most reactive metals are those from Groups 1 and 2. The ones place of the group number is the number of valence electrons in an atom of these elements. Generally, elements in Groups 1, 2, and 13 to 17 tend to react to form a closed shell with a noble gas electron configuration ending in #ns^2 np^6#. The Group number of a non-transition metal can be used to find the number of valence electrons in an atom of that element. 
Elements whose atoms have the same number of valence electrons are grouped together in the Periodic Table.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
